ORCHIDOMICS
Understanding the metabolism of orchids in their environments - omics methods unravel adaptations and symbioses in orchids
Research project funded by the National Science Centre (Poland) in MAESTRO 7 programme
The Beneficiary: University of Gdańsk
Principal investigator: Professor Marc-André Selosse, PhD
Summary
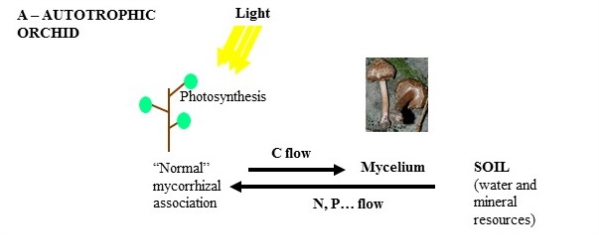
Orchids are very diverse in species number, but also in types of nutrition. Species that rely on photosynthesis in adulthood strikingly germinate underground from very tiny seeds without reserves: at germination, a fungus colonizing them provides all the nutrients required, a nutrition called mycoheterotrophy. Later in development, the plant eventually becomes green and photosynthetic. Yet, the fungus remains associated with the roots, now exchanging mineral resources collected in the soil against plant carbon. Orchids are thus important models when seeking to understand how nutrition and microbial interaction change during plant development.
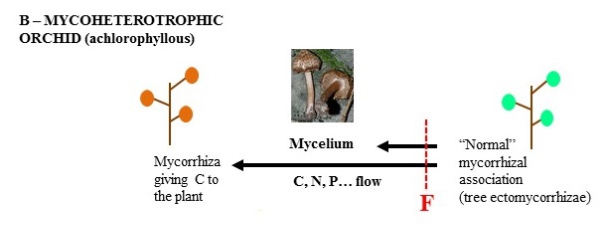
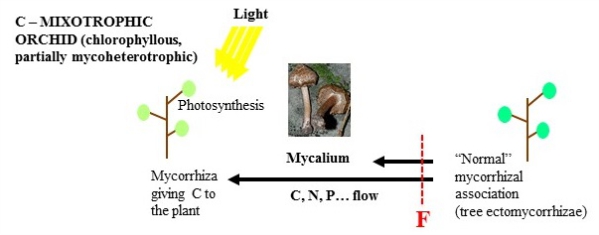
Interestingly, some orchids remain more or less mycoheterotrophic in adulthood. Some are not green and not photosynthetic: the fungus present in the roots provides them with all the nutrients required, allowing mycoheterotrophy in adulthood. Some species display an intermediate strategy: they are photosynthetic, but receive part of their carbon from the fungus, in addition to mineral resources, enabling them to adapt to living in light-limited forests (mixotrophy). These strategies also exist in other plants, but evolved often in orchids, which are thus interesting models when seeking to understand the mechanisms and evolution of the loss of photosynthesis in plants.

Finally, many orchids are rare or endangered species whose presence is an indicator of ecosystem health, so knowledge of their nutrition and relation to supporting fungi is crucial. Paradoxically, some species live in soils polluted, for example, by heavy metals, as some South Poland populations investigated here, although we do not know if they achieve there a normal physiological status.
ORCHIDOMICS aims at better understanding of these different types of nutrition and the role of the associated fungi, using orchids as models. It concentrates on underground germination, adult roots that interact with fungus, and adult photosynthetic leaves, in orchid species with various types of adult nutrition.
ORCHIDOMICS makes use of the most recent methods, which provide a lot of data from a single sample. First, the characterization of RNAs shows which plant genes are active. Second, extraction and separation of the main molecules (metabolites) provide a view of metabolism in action. The thousands of pieces of information gathered will be handled using computer analyses and statistics to (i) separate relevant data from noise, (ii) collate metabolic and gene expression data, and (iii) compare data obtained from different samples. The handling of unusually large datasets is the first challenge of ORCHIDOMICS. The second challenge is the study of orchids in situ: ORCHIDOMICS is based on limited, non-lethal sampling of orchid parts in natural populations, authorized by sampling permits. Thus, the data will reflect orchid physiology in living conditions.
In more detail, ORCHIDOMICS will investigate the diversity of orchid nutrition types. First, we will compare adult plants from nine species that are fully photosynthetic, partially mycoheterotrophic or fully mycoheterotrophic in adulthood. Second, we will monitor metabolic changes between mycoheterotrophic seedlings and adults in these species. For four species, we will also monitor the modifications produced by two environmental factors: light level and soil pollution by heavy metals.
ORCHIDOMICS will produce a massive dataset that will build a detailed view of the various orchid nutrition types in situ. This will first serve as a unique model to understand how plant nutrition changes with developmental stage, environmental conditions, and during evolution. Second, through better knowledge of orchid biology in realistic conditions, ORCHIDOMICS will help improve their future protection. Finally, it will pave the way to future, more precise analyses of crucial metabolites and genes involved in the different types of nutrition of orchids, and indeed ORCHIDOMICS itself encompasses more genetically detailed analyses.
Publications
| Selosse MA, Charpin M, Not F. Mixotrophy everywhere on land and in water: the grand écart hypothesis. Ecol Lett. 2017 Feb;20(2):246-263. | |
| Selosse MA, Minasiewicz J, Boullard B. An annotated translation of Noël Bernard's 1899 article 'On the germination of Neottia nidus-avis'. Mycorrhiza. 2017 Aug;27(6):611-618. | |
| Jacquemyn H, Waud M, Brys R, Lallemand F, Courty PE, Robionek A, Selosse MA. Mycorrhizal Associations and Trophic Modes in Coexisting Orchids: An Ecological Continuum between Auto- and Mixotrophy. Front Plant Sci. 2017 Aug 29;8:1497. | |
| Selosse MA, Schneider-Maunoury L, Martos F. Time to re-think fungal ecology? Fungal ecological niches are often prejudged. New Phytol. 2018 Feb;217(3):968-972. | |
| Strullu-Derrien C, Selosse MA, Kenrick P, Martin FM. The origin and evolution of mycorrhizal symbioses: from palaeomycology to phylogenomics. New Phytol. 2018 Mar 24. | |
| Tešitel J, Těšitelová T, Minasiewicz J, Selosse MA. Mixotrophy in Land Plants: Why To Stay Green? Trends Plant Sci. 2018 Aug;23(8):656-659. | |
| Lallemand F, Logacheva M, Le Clainche I, Berard A, Zheleznaia E, May M, Jakalski M, Delannoy E, Le Paslier M, and Selosse M-A. Thirteen New Plastid Genomes from Mixotrophic and Autotrophic Species Provide Insights into Heterotrophy Evolution in Neottieae Orchids. Genome Biol Evol 2019 Sep;11(9):2457-2467. | |
| Lallemand F., May M., Ihnatowicz A., Jąkalski M. The complete chloroplast genome sequence of Platanthera chlorantha (Orchidaceae). Mitochondrial DNA Part B-Resources 2019 4(2): 2683-2684. | |
| May M, Novotna A, Minasiewicz J, Selosse M-A, Jąkalski M. The complete chloroplast genome sequence of Dactylorhiza majalis (Rchb.) P.F. Hunt et Summerh. (Orchidaceae). Mitochondrial DNA Part B-Resources 2019 4(2): 2821-2823. | |
| Lallemand F, Figura T, Damesin C, Fresneau C, Griveau C, Fontaine N, Zeller B, Selosse M-A. Mixotrophic orchids do not use photosynthates for perennial underground organs. New Phytol. 2019 Jan;221(1):12-17. |
Gallery

Cephalanthera damasonium - albino specimen (by J. Gołębiewska)

Cephalanthera damasonium - mixotrophic orchid (by J. Gołębiewska)

Neottia nidus-avis - mycoheterotrophic orchid (by A. Kuczyńska)

Epipactis atrorubens - mixotrophic orchid (by J. Gołębiewska)

Platanthera bifolia - autotrophic orchid (by J. Gołębiewska)

Epipogium aphyllum - rare mycoheterotrophic orchid (by E. Krawczyk)

Fieldwork, Trzebinia (by A. Robionek)

Fieldwork, Miechowska Highland (by A. Kuczyńska)

sowing orchid seed "in situ" (by J. Minasiewicz)

Inocybe - species of this genus form symbiotic relationships with Epipogium aphyllum (by J. Gołębiewska)

Lecture at the "Orchid Festival" in Kalina Mała 05.2017 (by M. Jąkalski)

Samples collection stand in the field (by M. Jąkalski)

Sowing orchid seeds in nylon bags (by M. Jąkalski)

Checking the stage of seeds germination in the field (by M. Jąkalski)
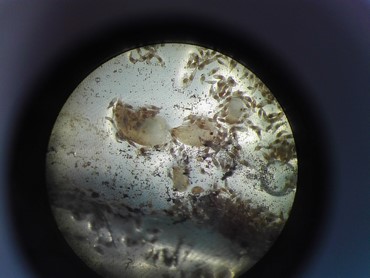
Germinating orchid seeds (protocorms) in nylon bag (by M. Jąkalski)
created by using html5up.net technology